Live blood checks - part 1
(microscopy of live blood in the nano-age)
Over the last six weeks or so I have visited communities north and south of Brisbane and looked at live blood samples on over 120 people. It is clear that the blood has got worse in the last three months.
Lots to discuss and if I try and do it all at once I won’t get finished so here goes with part one. There will be a part 0.5 discussing slide preparation and sampling technique but will do that later as well.
Firstly, as I have stated previously, this is different from live blood analysis which has its own history, nomenclature and devotees. I respect the work that has been done over a long period of time and will re-visit this also.
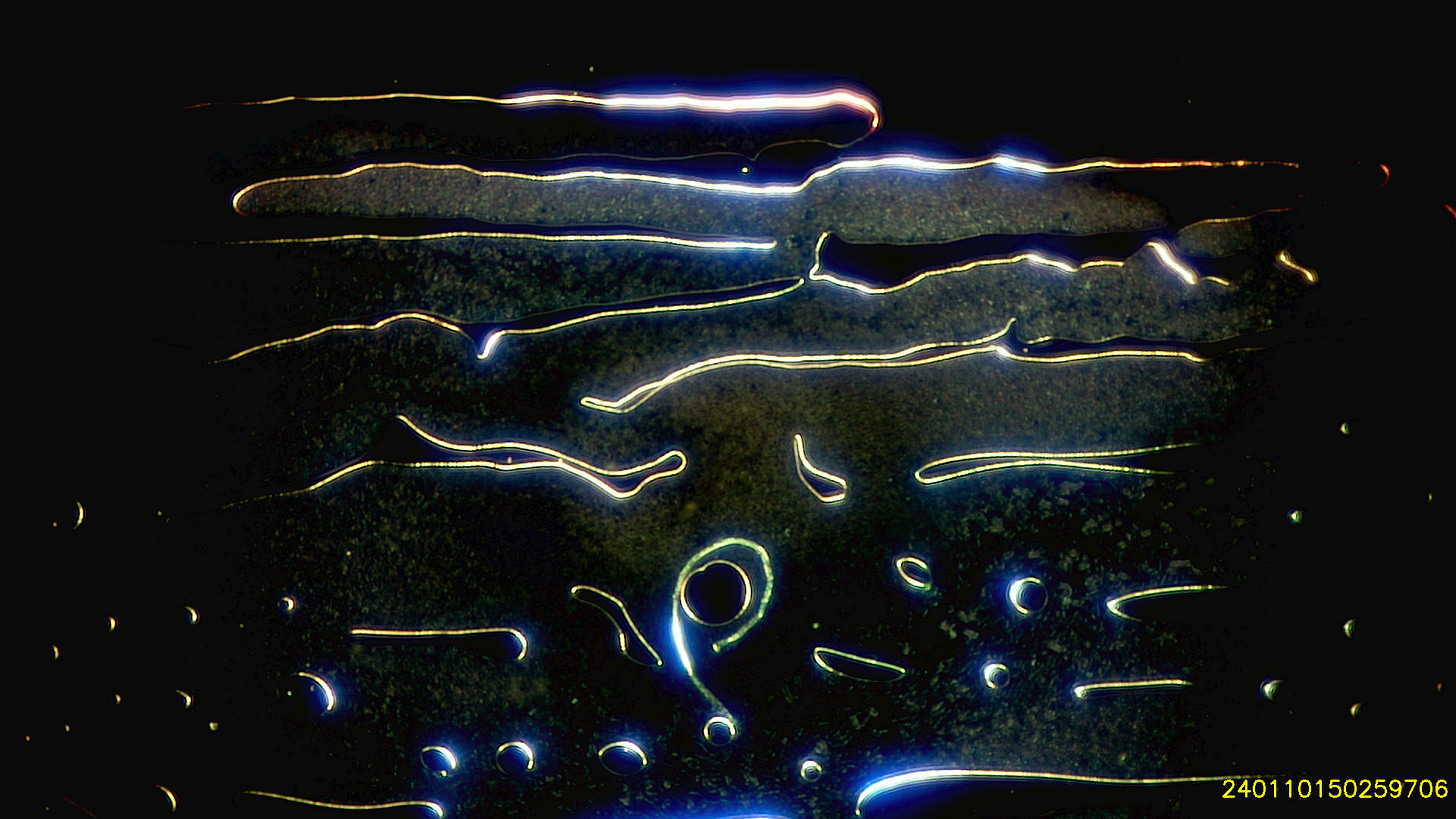
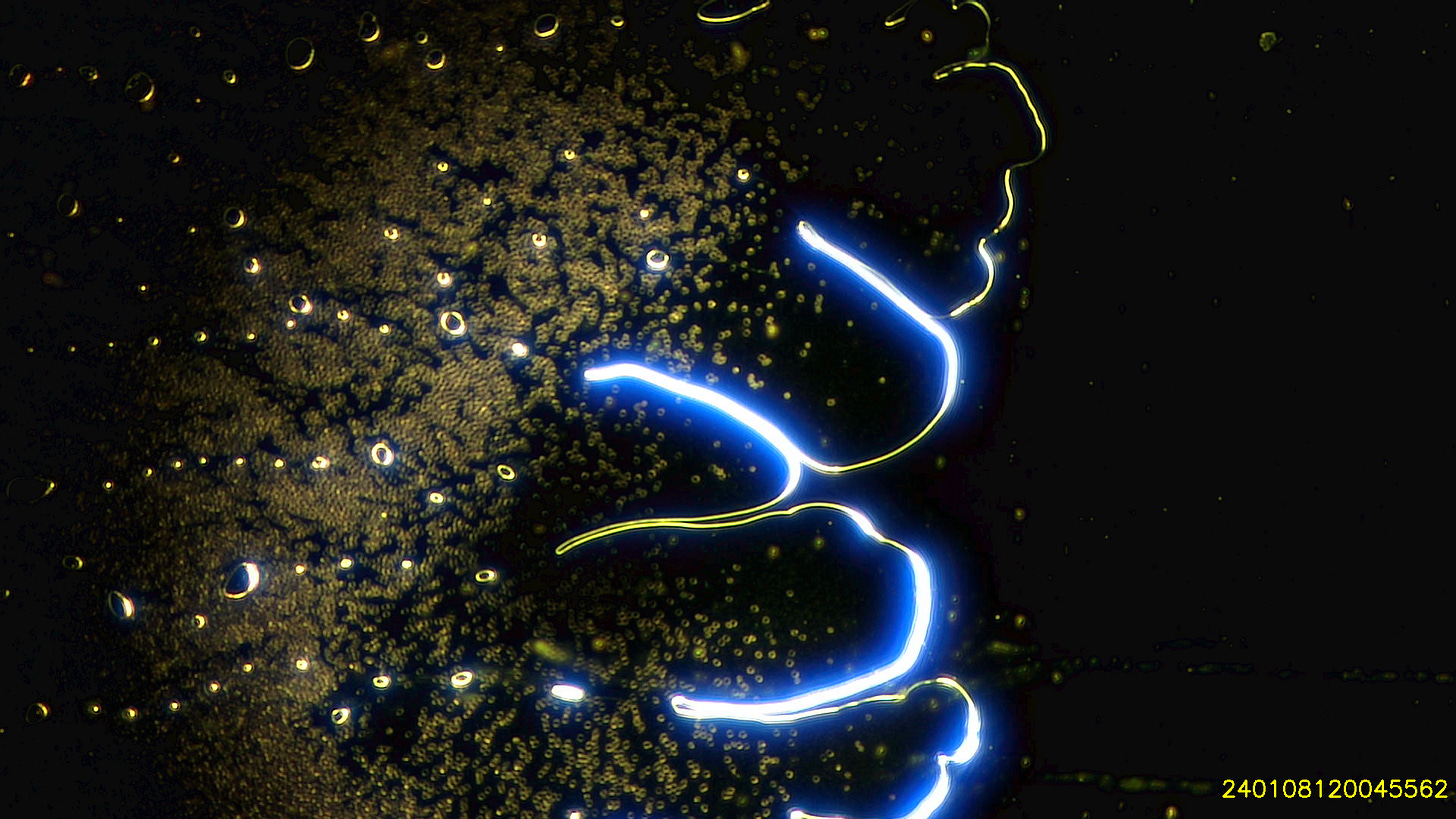
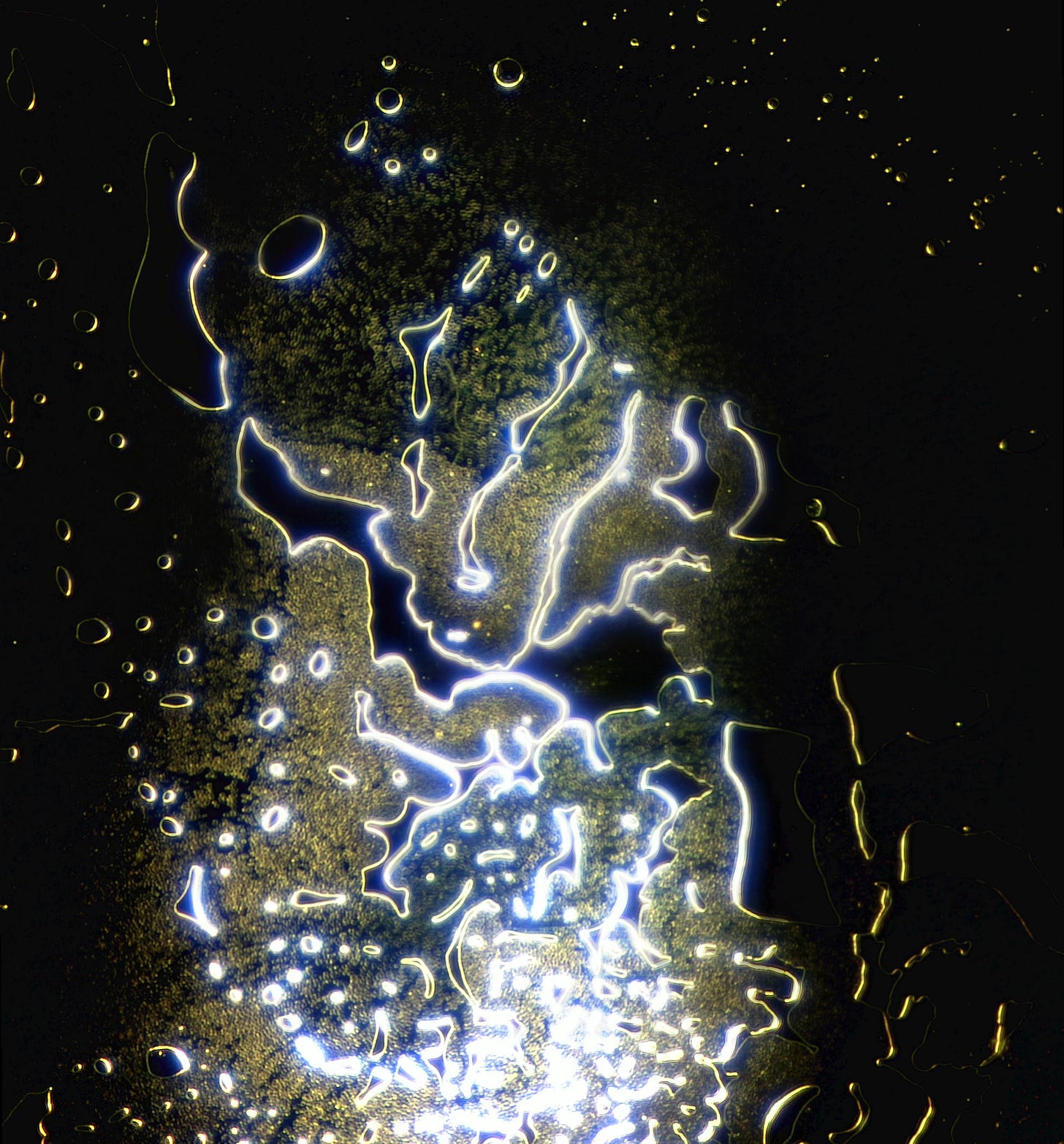
I use a small drop of blood which spreads over the slide under the coverslip. Even with the naked eye its possible to see that the blood no longer spreads like it used to. Most samples are moderately to severely contaminated with hydrogel and particles - which my colleagues and I refer to as ‘dots’, because we don’t know what they are or even how big they are. Except Shimon who prefers particles to dots.
Whilst 12 months ago I saw small bubbles in the blood - I now see hydrogel with holes in it around the edges and large hydrogel bubbles centrally.
Here are a few examples of hydrogel on the edges. Darkfield, most images are using the 2.5x objective, some the 10x.
Here are a few videos which show the hydrogel flowing. In the first one note the dots/particles that are ‘flying’ ahead of the red blood cells.
Hopefully that has given you a flavour as to how the hydrogel flows. In the next part I will take a look at the hydrogel bubbles near the centre of the sample, the ‘dots’, the structures, the neutrophils and a re-look at the effects of sodium citrate and activated charcoal. Following that I have been sent further water samples to evaluate and I aim to prioritise these. That is the plan. Will see how the week goes.
I hope yours starts well as we close in on the end of the first month of 2024.
David
PS For the most expensive coffees on Substack please visit;










































Dr Nixon ...
(SUBLINGUAL EDTA )
under the tongue IS having amazing results by people
It’s often confused
with ORAL EDTA Which is not effective as oral edta is destroyed in the stomach & also effects the stomach biome ..
BUT ..Simply emptying a 1000mg capsule of EDTA or 1000mg scoop of EDTA held under the tongue held for 6mins to dissolve directly into the blood stream cleans the blood 🩸 very efficiently as most people cannot afford
the expensive IV EDTA..
up to 60% of a 1000mg held under the tongue gets into the blood stream
& so much easier than sitting for a IV
it’s bizarre to me why this has been overlooked ..
I’m sure microscopy would show (incredible results)
looking at the blood before & after
sublingual Edta
Doing 1000mg EDTA twice a day
Would place the same amount of EDTA directly in the bloodstream as IV..
Your thoughts on this would be greatly appreciated as edta can be bought by the bucketload for penny’s .
Making it obtainable for all
Thank you Dr David Nixon
Hi Dr Nixon - when you're re-analysing effects of sodium citrate & activated charcoal, maybe try some horseradish peroxidase (HRP) as well. See this March 2011 paper -
"The enzymatic oxidation of graphene oxide" https://pubmed.ncbi.nlm.nih.gov/21344859/ - The paper states: "In the presence of low concentrations of hydrogen peroxide (∼40 μM), HRP catalyzed the oxidation of graphene oxide, which resulted in the formation of holes on its basal plane." I came across the paper in this substack article - https://siriusispodstruje.substack.com/p/graphene-oxide-detoxification Maybe you or someone here has tried it already?